
UDI – Mandatory Data Transmission to Anvisa’s Unique Device Identification System (SIUD) Begins March 1st
Anvisa’s Unique Device Identification System (SIUD) becomes operational this March. Normative Instruction (IN) 426/2026, which regulates the submission of information to the system, was published on February 13th in compliance with RDC nº 591/2021. This measure aligns Brazil with the standards defined by the International Medical Device Regulators Forum (IMDRF), directly impacting manufacturers, importers, and the distribution chain. The goal is to ensure the unambiguous identification of equipment and materials, enhancing patient safety and facilitating post-market monitoring (such as recalls and adverse events).
Unique Device Identification (UDI) uses an alphanumeric code for the exclusive and standardized identification of medical devices internationally. Already adopted in many countries, this identification brings significant advantages, increasing agility, safety, and reliability in medical care. It allows for fast and effective traceability, as well as transparency regarding the origin and applicability of supplies and devices.
The UDI code identifies the product and its manufacturing data, indicating the manufacturing unit and production site, serial and lot numbers, manufacturing date, and expiration date. Additionally, it identifies units within multiple-item packaging. For example, in a pack of 10 individual syringes, each one receives a separate identification.
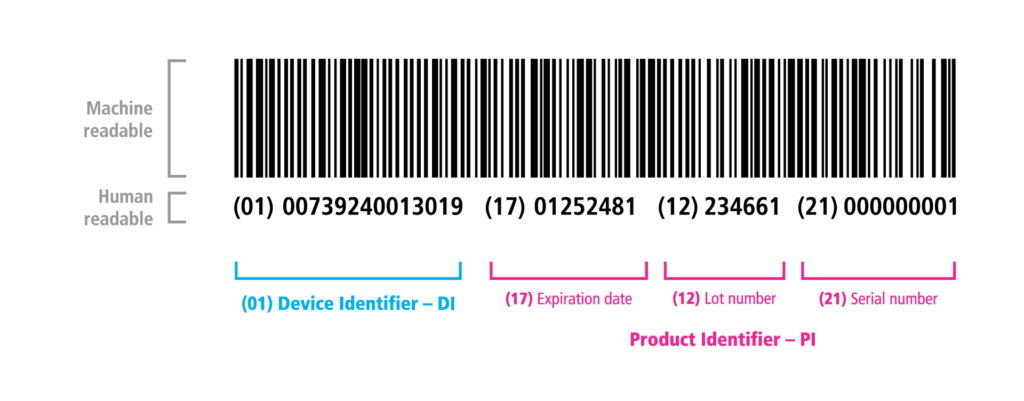
UDI barcode example with identification fields.
The UDI code is generated by recognized entities mentioned in Art. 11 of RDC 591/21 (GS1, HIBCC, and ICCBBA), which issue individualized barcodes and QR codes for the products.
What is the importance of UDI and its benefits?
The implementation of UDI is not merely a bureaucratic requirement but rather a competitive tool. Standardization favors access to foreign markets and strengthens the national industry, acting as a “passport” for exports by aligning Brazilian products with the requirements of international buyers.
For distributors and hospitals, the system allows for greater transparency in inventory management and faster procurement, in addition to facilitating the identification of products in medical records.
Furthermore, UDI improves communication between manufacturers and regulators. When a device presents an issue, it is easier to act quickly, which can prevent public health risks.
Unique identification also facilitates product management, as manufacturers can better control inventory and ensure devices are up to date, helping to prevent the use of outdated or unsafe products. Additionally, in cases of lost or stolen goods, it is possible to track them individually by scanning the codes, thereby inhibiting illegal practices.
What are the deadlines for UDI usage and transmission to the ANVISA system?
Anvisa’s mandatory Unique Device Identification (UDI) applies to all medical devices and IVDs (in vitro diagnostics) marketed in Brazil. The deadlines were staggered by RDC No. 591/2021 as follows:
Mandatory Requirement (Labeling/Packaging Marking):
- Class IV (Maximum Risk) – July 10, 2025
- Class III (High Risk) – January 10, 2026
- Class II (Medium Risk) – January 10, 2027
- Class I (Low Risk) – January 10, 2028
It is important to note that the deadlines for the mandatory transmission of data to the Unique Device Identification System (SIUD) differ from physical marking. These were established by IN No. 426/2026, which came into effect on March 1st:
- Class IV (Maximum Risk) – September 1, 2029 (3.5-year term)
- Class III (High Risk) – March 1, 2030 (4-year term)
- Class II (Medium Risk) – March 1, 2031 (5-year term)
- Class I (Low Risk) – March 1, 2032 (6-year term)
For guidance on this matter, please contact WeRegister through our service channels.

UDI – Mandatory Data Transmission to Anvisa’s Unique Device Identification System (SIUD) Begins March 1st

WeRegister na Revista NewsLab

Detenha seus próprios registros na Anvisa

Maximize suas oportunidades de mercado!

